Trusted by the world’s best
Engineered for Pharmaceutical Teams
Medical Writers and Regulatory Professionals
Spend less time on routine drafting and more on scientific quality — with AI that handles synchronization, formatting, and consistency checks automatically.
Regulatory Affairs and R&D Leaders
Accelerate IND, NDA, BLA, and MAA submissions with end-to-end pharma R&D document automation across the full drug development lifecycle.
IT and Digital Innovation Teams
Deploy a GxP-compliant AI writing platform for life sciences with native Microsoft 365 integration, Veeva API connectivity, and 21 CFR Part 11-compliant audit trails.
Automate Regulatory Authoring Across the Full Drug Development Lifecycle
Streamline the Submission Lifecycle
- End-to-End R&D Document Automation
- Comprehensive eCTD Coverage
- Faster IND, NDA, BLA & MAA Filings
AI-Powered Regulatory Authoring
- Drafts Grounded in Your Own Verified Regulatory Documents
- Cross-eCTD Document Network and Linkage Across Modules 1 to 5
- Content Change Orchestration Across Linked Documents and Markets
- End-to-end QC and Review
- Continuous Audit Readiness with 21 CFR Part 11-Compliant Audit Trails
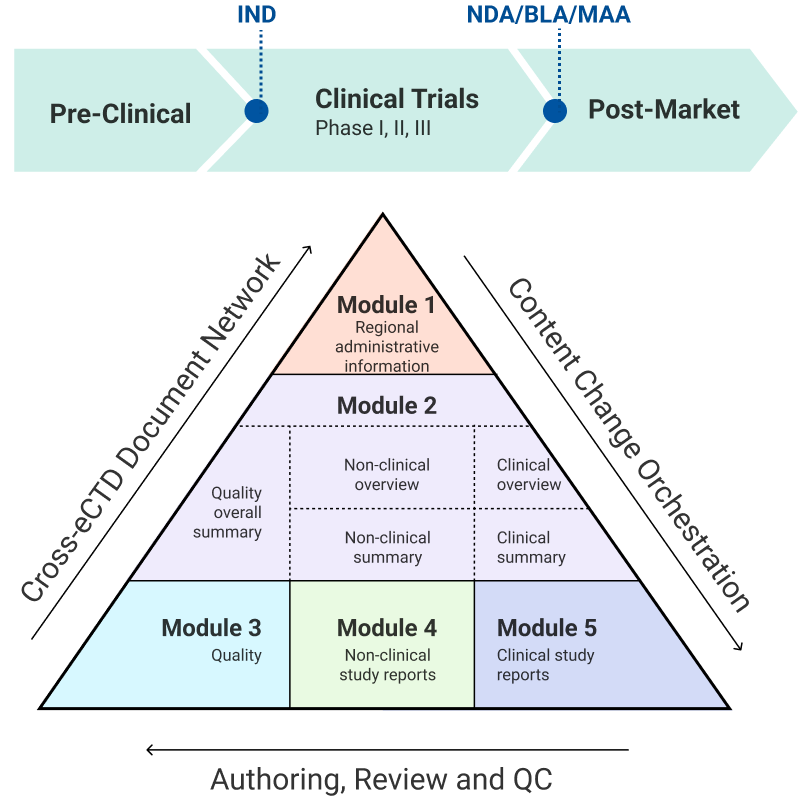
Accelerate Authoring Across All Core Regulatory Dossiers
An out-of-the-box solution supporting comprehensive clinical, nonclinical, and CMC authoring to accelerate your path to submission.
Clinical
- Protocol & Amendments
- M5 Clinical Study Reports (CSR)
- M2.7.3 Summary of Clinical Efficacy
- M2.7.4 Summary of Clinical Safety
- M2.5 Clinical Overview
CMC
- M3 Quality Documentation
- M2.3 Quality Overall Summary (QOS)
Nonclinical
- M2.4 Nonclinical Overview
- M2.6 Nonclinical Written & Tabulated Summaries
- M4 Nonclinical Study Reports
Trusted by Pharma Companies and CROs Globally
AuroraPrime is the AI writing solution for pharmaceutical companies trusted by leading global pharma and CROs.
21 CFR Part 11-Compliant | ISO 9001 and ISO 27001 Certified | 92 to 317% Proven ROI
Customer Success
If we can accelerate our submission one day, that is an additional earning. The exchange from simulated to real data will completely transform the way pharma companies work with submissions — and we can only do that because we have your tool available.
Head of Regulatory Medical Writing, Leading Global Pharma
New White Paper Avaliable

Download the Frost & Sullivan Report: How Leading Pharma & CROs Are Scaling AI with Built-In Quality Control
Produced in collaboration with AlphaLife Sciences, this white paper from Frost & Sullivan captures exclusive insights from global industry leaders — including Heads of Biometrics and Medical Writing at top pharma companies and CROs, alongside experts from Microsoft and former FDA officials.
Download ReportAuroraPrime RMA
A Microsoft Word 365 add-in and AI platform for medical content authoring across the complete R&D document lifecycle, built for 21 CFR Part 11-compliant pharmaceutical workflows.
- Cross-Functional R&D Document Support Spanning Clinical, Nonclinical, CMC, Safety, and Post-Market
- Configurable eCTD Templates with Content Reuse Across Modules and Markets
- IND, NDA, BLA, and MAA Filing Acceleration from Initial Drafting to Final Review and Submission
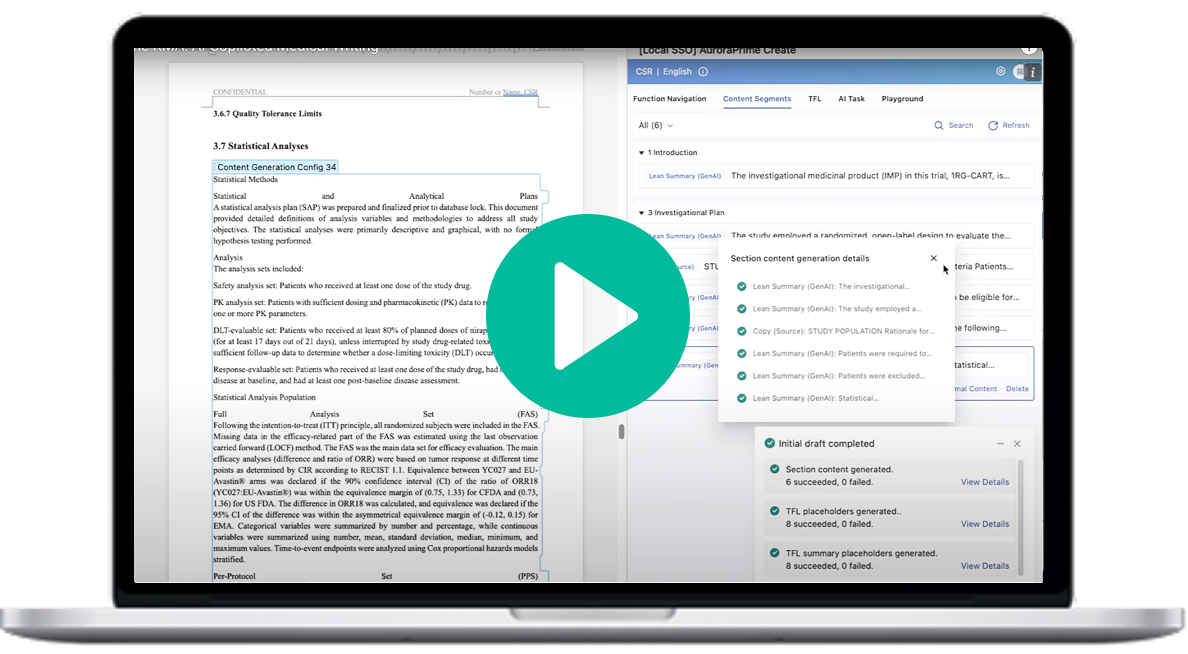
Products & Services
AuroraPrime RMA
AI-Powered Regulatory Authoring
Accelerate R&D documentation with a scalable, compliant AI platform. RMA grounds drafting in your verified regulatory content, integrates natively with Microsoft Word and Veeva Vault, and keeps medical writers in control from first draft to final approval.
Explore AuroraPrime RMAAuroraPrime DocMind
Enterprise AI Engine for Pharma
Empower pharma IT to build, scale, and orchestrate enterprise AI content and data solutions. A composable architecture lets internal teams manage templates, workflows, and integrations independently — purpose-built for regulated life sciences, not retrofitted from generic AI.
Explore DocMindAuroraPrime Expert
AI-Accelerated Authoring Services
Deliver submission-ready documents faster and to enterprise-grade standards. AlphaLife Sciences pairs AuroraPrime automation with senior medical writing and regulatory expertise — applying the same human-in-the-loop principle to your most time-critical documents.
Explore Expert ServicesBuilt for Enterprise Trust and Compliance
AuroraPrime is built for the compliance, security, and validation standards that regulated pharma R&D demands. Our pharma R&D document automation solution operates within 21 CFR Part 11, HIPAA, GDPR, SOC 2 Type II, ISO 9001, and ISO 27001 frameworks, with adherence to global standards.
Latest News
AlphaLife Sciences and Veeva Finalize Strategic Product Partnership
AlphaLife Sciences finalizes its strategic product partnership with Veeva, enabling seamless Veeva Vault RIM integration for AI-orchestrated regulatory authoring.
May 06, 2026AlphaLife Sciences Announces Veeva RIM Integration to Power AI-Orchestrated Authoring and QC
AlphaLife Sciences Announces Veeva RIM Integration to Power AI-Orchestrated Authoring and QC
Mar 30, 2026Microsoft x AlphaLife: Powering Regulatory-Compliant AI Workflows with PostgreSQL on Azure
🚀 What happens when enterprise-grade cloud infrastructure meets domain-specific AI for life sciences?In our latest collaboration with Microsoft, we demonstrate how PostgreSQL on Azure can power secure, high-performance AI pipelines designed specifically for the stringent requirements of the life sciences industry. The result: faster insights, stronger compliance, and AI workflows that are ready for real-world regulatory environments.From structured regulatory data to intelligent document workflows, this partnership shows how modern cloud databases and AI-driven platforms can work together to unlock a new era of operational efficiency and scientific productivity.If you're exploring how AI and cloud infrastructure can reshape regulatory operations, this is a story worth reading.
Mar 10, 2026Frequently Asked Questions About AlphaLife Sciences

Newsletter Sign Up
Subscribe to our newsletter to receive the latest updates and insights on AI in life sciences.


























