Accelerating Clinical Trial Protocol and Protocol Amendments with AI
Apr 01, 2026Rethink how your team handles Protocol & Protocol Amendments. AlphaLife Sciences' AuroraPrime RMA cuts down drafting hours and tightens compliance across the board.
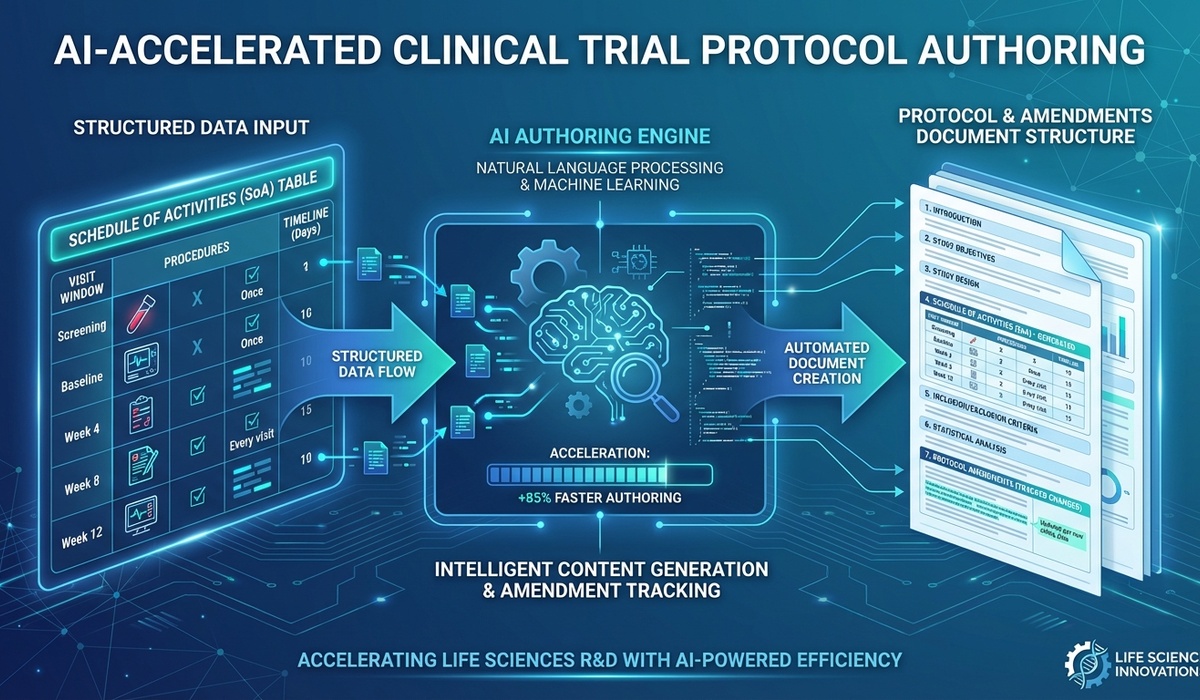
The backbone of any clinical trial is its design. That design lives inside the clinical study protocol. Yet, writing a solid protocol—and tackling those inevitable Protocol & Protocol Amendments—is still a massive time-sink for drug development teams.
Medical writers regularly lose hundreds of hours cross-checking conceptual sheets. They try to keep data straight across massive tables and have to manually update dozens of sections the moment a single parameter shifts.
What if you could win that time back?
An AI Writing Solution for Life Sciences changes the game entirely. AlphaLife Sciences’ AuroraPrime RMA platform is reshaping how organizations handle protocol creation, effortlessly closing the gap between study design and getting that final regulatory nod.
Here is what we'll cover:
Real bottlenecks in manual protocol authoring
How AI drafts and populates complex study sections for you
Why automated Schedule of Assessments (SoA) extraction changes everything
Smart tactics to future-proof your Protocol & Protocol Amendments
Let’s look at how AI Regulatory and Medical Authoring can speed up your path to “First Patient In.”
Overcoming Bottlenecks in Protocol Authoring
Writing a study protocol isn't a straight line. It takes constant back-and-forth across clinical operations, biostatistics, and other functional teams. When you rely completely on manual processes, you quickly run into a few big roadblocks.
The Problem with Data Consistency
A clinical protocol needs perfect consistency. Change one study parameter, and the ripple effect hits everything from the synopsis to the statistical plan. During Protocol & Protocol Amendments, spending hours hunting down and changing these scattered data points manually is a huge driver of regulatory delays and QA findings.
Losing Time to Administrative Tasks
Incredibly skilled medical writers spend way too much time doing formatting chores. They copy and paste boilerplate text. They fight with Microsoft Word's formatting rules. That leaves them less time to focus on what matters: scientific rigor and the safety of participants.
Pro Tip: The most successful regulatory teams see their medical writers as scientific strategists. They offload the repetitive, manual drafting work to an AI system.
Generating Protocols with AuroraPrime RMA
AuroraPrime RMA shakes up the old workflow by bringing smart automation straight into Microsoft Word. Think of it as an advanced co-author that natively speaks the language of regulatory frameworks.
Drafting Right from the Concept
Instead of staring down a blank template, writers can instantly generate complete first drafts. They just instruct the AI to reference existing clinical study conceptual sheets or protocol synopses. The system scans the source documents, pulls out the vital study designs, objectives, and endpoints, and maps them directly into your standard protocol templates.
Automating SoA Table Extraction
If there's one standout feature in AuroraPrime RMA for Protocol & Protocol Amendments, it's how it handles the Schedule of Assessments/Activities (SoA) table.
The SoA table is the core of any protocol. RMA takes this massive matrix and instantly turns it into a machine-readable object. From there, it automatically generates written sections like "Demographics and Baseline Characteristics" or "Study Assessments and Procedures." We're talking minutes, not days.
| Traditional Manual Protocol Authoring | AuroraPrime RMA AI-Assisted Authoring |
|---|---|
| Manual cross-referencing of SoA data | Automated data flow from SoA to text |
| Days spent mapping conceptual texts | Minutes to generate initial draft |
| High risk of versioning inconsistencies | System-traced source grounding |
Creating a Connected Ecosystem
A standalone tool for AI writing is nice. A fully connected platform changes everything. AuroraPrime RMA includes native Veeva Vault RIM Integration. This means your source documents, custom templates, and working drafts all sit in a validated, secure environment.
Every single piece of evidence the AI uses to help draft your Protocol & Protocol Amendments links directly back to its original source. You keep an unbroken chain of custody for compliance without even trying.
Frequently Asked Questions
How does AI handle complicated Protocol & Protocol Amendments?
When a protocol simply has to change, writers ask AuroraPrime RMA to look at specific sections. They give new instructions or point to updated literature, and the AI regenerates just the pieces that need updating, keeping full traceability to the new reference material.
Is AI-generated protocol content secure?
Yes. AlphaLife Sciences designed these platforms exclusively for life sciences. That means enterprise-grade security and total data segregation. Your proprietary clinical data is never used to train public AI models.
Can the AI system adopt our company’s specific writing style?
Absolutely. AuroraPrime RMA lets you load up sponsor templates and set detailed generation rules. You can tell the AI to use your organization's exact voice, preferred formatting, and specific regulatory setups.
Wrapping Up
The days of slow, labor-intensive clinical protocol writing are fading out. By bringing in AI Regulatory and Medical Authoring solutions like AuroraPrime RMA, your team can handle Protocol & Protocol Amendments with speed, unmatched accuracy, and tighter consistency.
When you cut out the administrative drag of mapping SoA tables and wrestling with document logic, you empower your medical writers to elevate the science behind your clinical programs.
Ready to accelerate your clinical trial submissions? See what's next in regulatory writing and explore how AuroraPrime RMA can change the way you work right now.





