Accelerating CTD Module 5 Clinical Study Reports (CSR) with AI
Apr 09, 2026Discover how an advanced AI Writing Solution for Life Sciences like AuroraPrime RMA can speed up your CTD Module 5 Clinical Study Reports (CSR) authoring process.
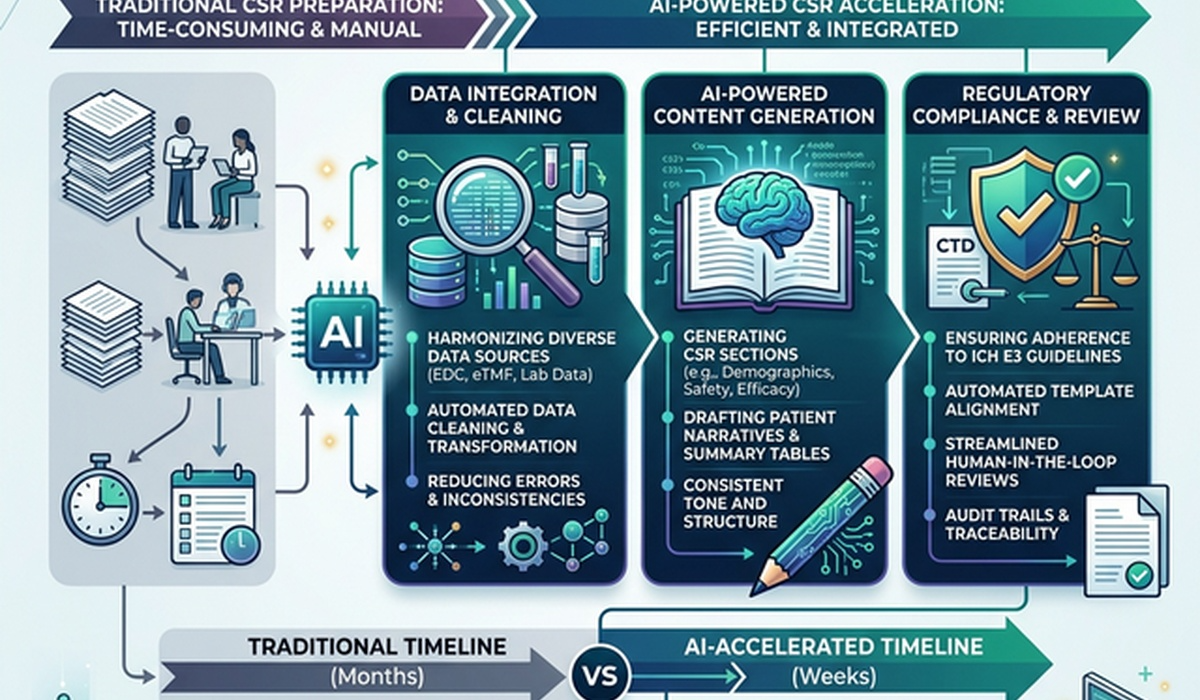
Writing a regulatory-compliant clinical study report really doesn't need to feel like a constant race against the clock.
If you're a medical writer or working in regulatory affairs, you know the drill all too well. Finalizing a CTD Module 5 Clinical Study Reports (CSR) usually means late nights, endless manual data integration, meticulous formatting checks, and the ever-frustrating task of updating Tables, Figures, and Listings (TFLs). With clinical trials growing more complex and data volumes exploding, sticking to the traditional manual way of authoring just slows down regulatory submissions—and frankly, it opens the door for more errors.
But imagine if you could hand off the most tedious, repetitive parts of writing a CSR without losing an ounce of scientific rigor.
In this article, we’re going to dive into how bringing in an advanced AI Writing Solution for Life Sciences—like AuroraPrime RMA—is completely rethinking the CSR authoring process. We’ll look at how things like AI-guided templates, automatic TFL integration, and smart summarization are actually helping teams cut down document cycle times, save money, and make clinical trial submissions much smoother.
The Real Bottleneck in CTD Module 5 Clinical Study Reports (CSR) Authoring
Putting together a high-quality CTD Module 5 Clinical Study Reports (CSR) means wrestling with massive chunks of data from protocols, statistical analysis plans (SAPs), and biostatistics outputs. For most teams, the biggest headache usually comes down to managing those TFLs.
Medical writers are constantly battling:
Fragmented Data Sources: Trying to manually stitch together data from hundreds of loose
.rtffiles into one coherent document.Data Synchronization Issues: Having to comb through and update the entire CSR just because of a late-stage statistical tweak.
Formatting and Display Constraints: Agonizing over complex, multi-page tables to make sure they meet rigid submission guidelines without resorting to manual, error-prone tweaks.
These repetitive, low-value chores can easily eat up to 70% of a writer's week. That leaves painfully little time for the high-value critical analysis and clear scientific storytelling that regulatory reviewers are actually looking for.
How an AI Writing Solution for Life Sciences Changes the Game
Bringing an AI Writing Solution for Life Sciences like AuroraPrime RMA into the fold moves the needle from manual heavy-lifting to automated generation and smart review. Because it lives right inside your familiar Microsoft Word 365 environment, AuroraPrime RMA tackles the biggest frustrations of CSR writing head-on.
AI-Enabled Document Templates
A great CSR starts with a solid first draft. With AuroraPrime, you kick off a new document using pre-built, AI-enabled templates (like the standard TransCelerate CSR template). By linking up your source documents—say, the study protocol and the SAP—the platform's Generative AI automatically pulls out and synthesizes the right context to build out those initial draft sections.
This completely wipes out the dreaded "blank page" syndrome and makes sure your objectives, study designs, and methodologies line up perfectly with your source truth from day one.
Batch Incorporating TFLs from Source
Let's face it: dealing with TFLs is notoriously painful. AuroraPrime RMA takes the sting out of it with a centralized TFL Source Data Directory.
Instead of mindlessly copying and pasting tables one by one, you can just batch-upload your TFL source files. From there, the system's AI Recommendation engine automatically matches those files to the right placeholders in your CSR. If your data stretches across multiple regional tables, you can use the Merge Tables tool to combine them smoothly into one submission-ready view—all without ever touching or skewing the underlying, verified data values.
Intelligent Data Synchronization and Summarization
When the biostats team hands over an updated cut of data, having to manually update your CSR is a recipe for transcription errors. AuroraPrime RMA’s Priority for TFL Content Sync steps in here. When updated TFLs are loaded into the system, your in-text tables are automatically refreshed with the newest data points, while keeping all your custom captions and formatting exactly how you left them.
On top of that, the platform uses Generative AI to churn out high-quality TFL Summaries. By giving it a simple custom prompt (like, "Summarize the key efficacy endpoints in 150 words"), the AI drafts clean, data-backed narrative text that interprets those freshly synced tables, saving you hours of writing time.
Frequently Asked Questions
What exactly is a CTD Module 5 Clinical Study Reports (CSR)?
A CTD Module 5 Clinical Study Reports (CSR) is a highly detailed, strictly structured document submitted to regulatory agencies (like the FDA or EMA). It gives them a comprehensive look at the design, execution, and results of a specific clinical trial.
How does AI keep data secure and accurate in CSRs?
An FDA-compliant AI Writing Solution for Life Sciences like AuroraPrime RMA protects data integrity by extracting text directly from your approved source documents (like SAPs) and programmatically pulling in locked TFL data. It lets writers tweak the view of the data (like merging rows for readability) without ever altering the verified numbers underneath.
Is AI going to replace medical writers?
Not at all. Think of AI as an incredibly capable assistant that takes the tedious formatting, copy-pasting, and initial drafting off your plate. It frees up medical writers to do what they do best: act as strategic editors and expert scientific communicators who focus on interpreting data and weaving a cohesive story.
Conclusion
Let's be honest—the old, manual way of writing a CTD Module 5 Clinical Study Reports (CSR) just isn't sustainable anymore, especially with trials getting more complex and submission deadlines getting tighter. By adopting a modern AI Writing Solution for Life Sciences like AuroraPrime RMA, regulatory teams can automate the heavy lifting of TFL integration, speed up their initial drafting, and ensure their data syncs flawlessly.
Ready to take your time back and focus on the science instead of the formatting? See for yourself how AuroraPrime RMA can reshape your clinical documentation process and help you hit those regulatory submissions faster and more accurately.





