Do Not Let Submission Failures Sink Your Clinical Trial: How AuroraPrime RMA Can Help
Mar 04, 2026A single submission failure can ripple across timelines, budgets, and patient access — and in today’s regulatory environment, there is zero margin for avoidable risk. 🚢📉What if your regulatory and medical authoring ecosystem could proactively surface inconsistencies, orchestrate cross-functional inputs, and generate submission-ready content with traceable intelligence built in?At AlphaLife Sciences, we believe AI should do more than accelerate drafting — it should fundamentally de-risk clinical development. 🚀 Through AuroraPrime RMA, we are redefining how regulatory, medical, and clinical teams collaborate, moving from fragmented document workflows to an integrated, AI-driven authoring framework designed for precision, compliance, and strategic control.
The pharmaceutical industry is no stranger to high costs. Developing a single drug can cost billions of dollars and take many years. Every day of delay can cost a company between $600,000 and $8,000,000. With stakes so high, it’s crucial to avoid any setbacks that could further inflate costs or delay timelines. One of the most significant risks? Submission failures. These failures not only cause delays, but also require additional resources to fix and resubmit.
The High Price of Submission Failures
Regulatory submissions are a crucial part of the drug development process. These documents, like Clinical Study Reports (CSRs), protocols, and safety narratives, need to be incredibly accurate and compliant. Even a small error or inconsistency can lead to rejection or requests for more information, causing expensive and time-consuming delays. Imagine the cost of reworking a document, not once, but multiple times. It’s a nightmare scenario that many companies face.
Enter AuroraPrime Regulatory and Medical Authoring (RMA)
What if you could significantly reduce the risk of submission failures and accelerate the entire process? That's where AuroraPrime RMA comes in. This powerful tool, seamlessly integrated with Microsoft Word 365, is designed to help medical writers produce high-quality, compliant documentation efficiently.
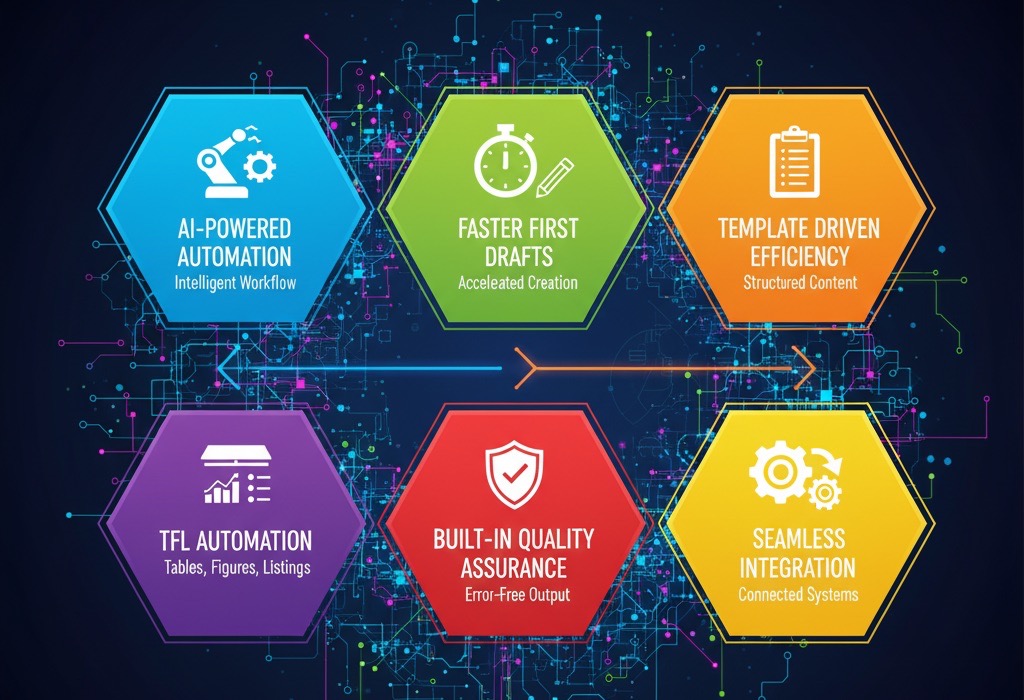
Here’s how AuroraPrime RMA can help you avoid costly submission failures:
AI-Powered Automation: AuroraPrime RMA uses generative AI to automate repetitive and time-consuming tasks. This includes generating first drafts, reusing existing content, and ensuring consistency across documents.
Faster First Drafts: The platform can generate "80% complete" drafts in minutes, significantly reducing document creation timelines. For example, first draft time for CSRs can be reduced by 90%.
Template Driven Efficiency: Use custom templates that are specific to therapeutic areas, ensuring consistency and accuracy. The system allows for easy template configuration, font styles, and built-in writing guidance.
Table, Figure, and Listing (TFL) Automation: The tool efficiently inserts TFLs into CSRs and keeps them up-to-date. It also generates summaries based on TFL data, reducing manual effort and ensuring regulatory alignment.
Built-In Quality Assurance: AuroraPrime RMA ensures compliance with regulatory standards and includes quality checks. Features such as text polishing ensure clarity and accuracy.
Seamless Integration: The add-in integrates seamlessly with Microsoft Word 365. It also integrates with other platforms such as RIM and Microsoft 365, further streamlining the workflow.
By automating these tasks, medical writers can focus on more strategic and value-added work, like data analysis and insights. This not only improves efficiency but also helps ensure that all documents are thoroughly reviewed and free of errors before submission.
Real Results and Tangible Benefits
AuroraPrime RMA isn't just a theoretical solution; it's delivering real results. The platform has been successfully piloted with top global pharmaceutical companies, and has been shown to reduce document production time.
Here’s a glimpse at the potential benefits:
CSR first draft time reduced by 90% and overall production time by 45%
Protocol first draft time reduced by 90% and total production time by 50%
Safety Narrative first draft time reduced by 95% and overall production time by 70%
With a reduction of this magnitude, companies can save significant amounts of time and money, and accelerate the drug development lifecycle.
Stop Wasting Time and Money on Submission Failures
Submission failures are a major obstacle in clinical development, but they don’t have to be. By leveraging the power of generative AI and automation, you can produce high-quality, compliant documents faster and more efficiently than ever before. With AuroraPrime RMA, you can:
Reduce the risk of submission failures.
Accelerate your document creation timelines.
Lower costs associated with manual documentation processes.
Focus on strategic tasks and innovation.
Bring life-saving treatments to market faster.
Are you ready to revolutionize your medical writing process and stop wasting valuable time and resources on submission failures? Contact AlphaLife Sciences today to learn more about how AuroraPrime RMA can transform your clinical trials.
By adopting AI-driven solutions like AuroraPrime RMA, you're not just streamlining workflows; you're investing in accuracy, compliance, and ultimately, success.





